|
However, these elements are reactive enough that they do not exist in their elemental forms in nature, but are present as compounds. The need to remove two electrons in order for the material to react means more energy is needed for electron removal. The Group 2 elements tend to be less reactive than their Group 1 counterparts. 
However, radium is a radioactive element and is generally under the category of radioisotopes in addition to being an alkaline earth metal, because it is not a stable element. Radium (atomic number 88) has similar properties to barium and is also in the Group 2 category. Mg has atomic number 12, this means it has 12 electrons. Ionization energy is defined as the amount of. Energy Sub-levels: A group of atomic orbitals within an energy level, that all have the same energy. Send us feedback about these examples.\) (Credit: Ingmar Runge Source: (opens in new window) License: Public Domain) Definition of energy levels (electron shells, shells) 1) The possible locations around an atom where electrons having specific energy values (quantum number) may be found. Metallic character refers to the level of reactivity of a metal to donate electrons during a chemical reaction. These examples are programmatically compiled from various online sources to illustrate current usage of the word 'energy level.' Any opinions expressed in the examples do not represent those of Merriam-Webster or its editors. Daniel Fienberg, The Hollywood Reporter, 13 Nov. Atomic radii increase down each group (column) of the periodic table because each successive energy level adds another layer of electrons, thereby making the atom larger. Metals are hard solid materials that have shine and other properties. 2023 The pictures are great, the anecdotes are fine and the general energy level is low. In chemistry, metals are defined by the properties such as luster, ductility, malleability, conductivity. Orbitals are generally drawn as three-dimensional surfaces that enclose 90 of the electron density, as was shown for the hydrogen 1 s, 2 s, and 3 s orbitals in part (b) in Figure 1.2.2. Rachel Crowell, Scientific American, 6 Dec. For a given atom, the s orbitals also become higher in energy as n increases because of their increased distance from the nucleus. 2023 Their objective was to explore whether band theory-which researchers use to consider the energy levels of materials and the atoms that they’re made of-could be reformulated to explain hyperbolic materials, which have irregular, warped arrangements. 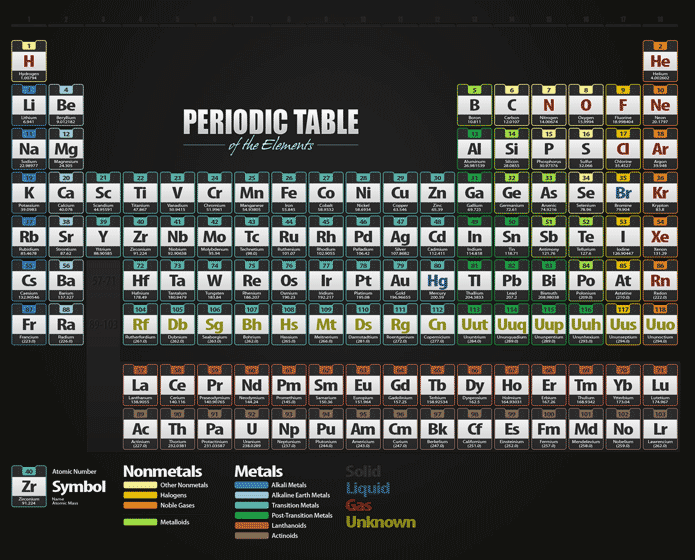
2023 This wellness journal allows students to track their sleep and energy levels plus set intentions for each day. His periodic law states that the chemical and physical properties of the elements vary in a periodic way with their atomic weights. 2024 Many of these products can make impressive health claims, marketing themselves as a quick way to transform gut health, improve immunity, boost energy levels, and even supercharge your metabolism. 2024 Pick one that fits with your schedule, energy level and sense of purpose. 2024 However, Garrison points out that forgoing alcohol for a week offers a glimpse into the upsides, namely improvements in physical and mental health such as better sleep, more mental clarity, and higher energy levels. Scientists use the periodic table to quickly refer to information about an element, like atomic mass and chemical symbol. In the periodic table the halogens make up Group 17 (according to the numbering system adopted by the International Union of Pure and Applied Chemistry), the group immediately preceding the noble gases. 2024 The green points to her creative mind, liveliness, and high energy level. transition metal, any of various chemical elements that have valence electronsi.e., electrons that can participate in the formation of chemical bondsin two shells instead of only one. The chemical behaviour of the halogen elements can be discussed most conveniently in terms of their position in the periodic table of the elements. Mandy Conrad, Discover Magazine, 10 Jan. The energy level can be defined as the stable or constant energy state achieved by a system consisting of particles like atoms, electrons, etc. It increases moving down a column or group and also increases moving from left. Electron affinity follows a trend on the periodic table. It is indicated using the symbol Ea and is usually expressed in units of kJ/mol. By November 2016, the International Union of Pure and Applied Chemistry had recognized a total of 118 elements. 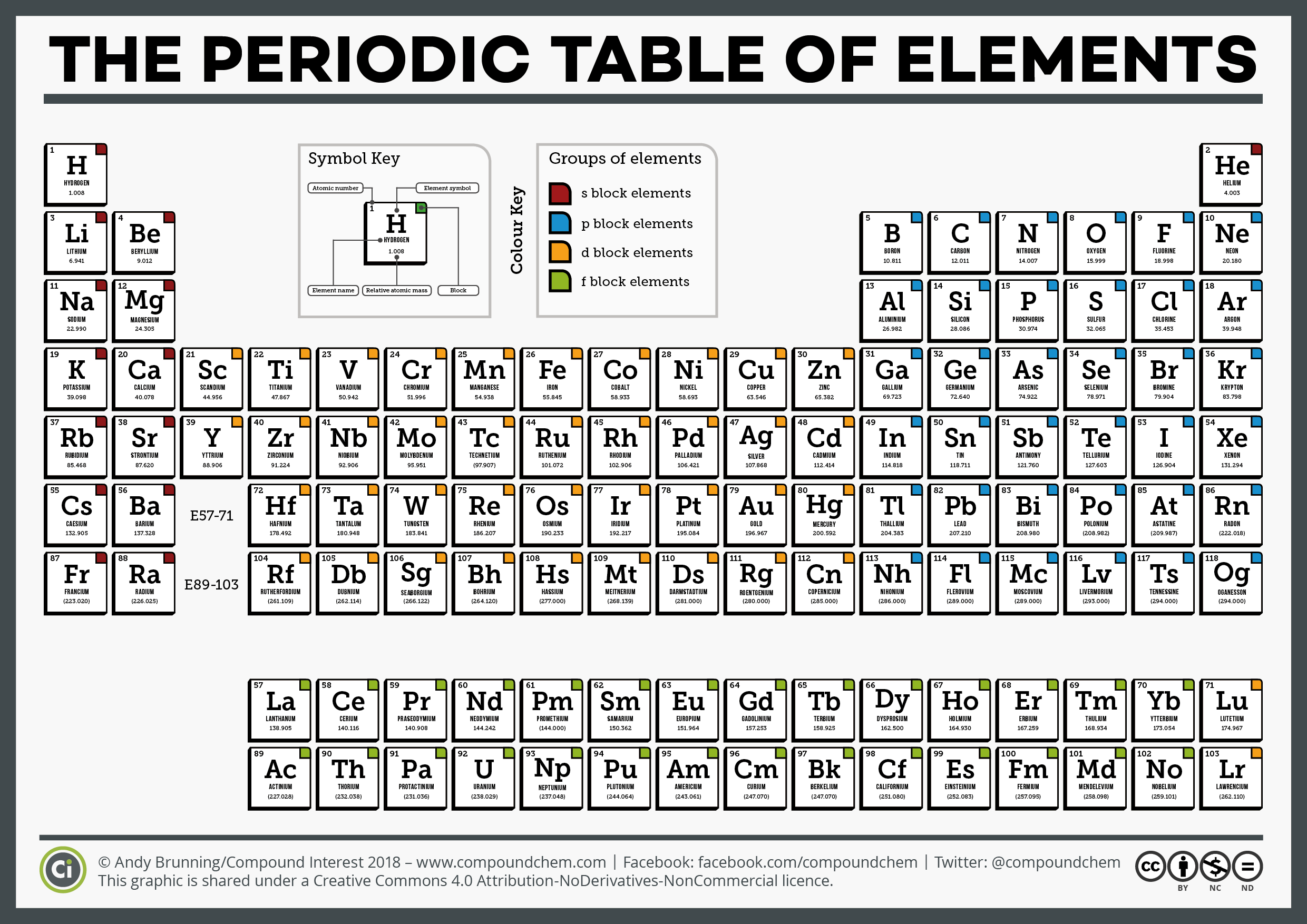
Recent Examples on the Web Meal timing and types of foods eaten can affect energy levels, satisfaction and hunger levels. Electron affinity is the amount of energy required to detach one electron from a negatively charged ion of an atom or molecule. The periodic table summarizes various properties of the elements, allowing chemists to derive relationships between them and to make predictions about compounds and potential new ones.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
